|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
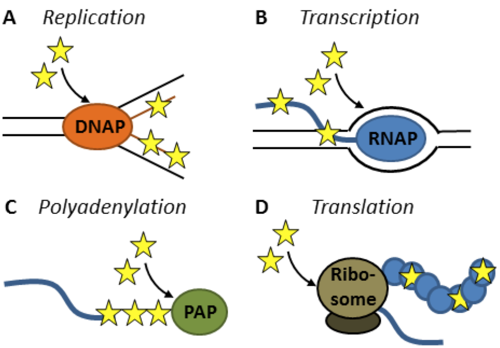
Metabolic labels offer straightforward approaches to directly address the three central pathways of molecular biology, namely replication, transcription and translation in life cells. Acting as analogues of natural biomonomers, metabolic labels are applied to their cellular target by simple feeding, being incorporated into the corresponding biopolymers by the usual enzymatic machinery. Combined with modern CLICK Chemistry, structural alterations induced by the label can be minimized, thereby permitting excellent enzymatic acceptance and mutually orthogonal labeling.
Figure 1: Scope of application for metabolic labels available from Jena Bioscience. Modified (deoxy)nucleosides are ideally suited to study the biosynthesis of nucleic acids. Once phosphorylated by cellular kinases, they undergo incorporation into (A) DNA by DNA polymerases (DNAP)[1] or into RNA by (B) RNA polymerases (RNAPs)[2] and (C) poly(A)-polymerases (PAPs)[3]. Similarly, labeled amino acids are substrates of cellular aminoacyl tRNA synthetase and thereby become available for protein synthesis (D). Modified puromycin derivatives offer an alternative for protein labeling in life cells[4].
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Responsible for content / Imprint | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Jena Bioscience GmbH
Jena Bioscience GmbH Loebstedter Str. 71 07749 Jena Germany Phone: +49 – 3641 – 6285 000 Fax: +49 – 3641 – 6285 100 E-Mail: info@jenabioscience.com Register Court: Amtsgericht Jena, HRB 207171 VAT No.: DE 195825742 Managing Directors: Thomas Billert Dr. Mathias Grün |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| www.jenabioscience.com | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||